Hydrogen is so light that the Earth’s gravity cannot hold it. It wanders up to the top of the atmosphere where it escapes like a molecule of steam coming off of boiling water, or where it is simply eroded away by the solar wind. Hydrogen loves to combine with other things though so there’s plenty of it bound at the molecular level. Most notably of course it is bound up with oxygen to make water.
Watts up, Doc?
All of the energy we use comes from hydrogen. Of course, it may have been created using hydrogen millions of years ago and sequestered somewhere (say as fossil fuels, or radioactive elements buried in the soil), but without hydrogen nothing else would exist.
Across Europe incoming solar energy (insolation) ranges between 2.75 and 5.44 kilowatts per square metre, annually. In North America that range falls between 2.87 and 5.89; in Australia, it runs from 4.17 – 6.46 kw/m². To put that all into perspective, the amount of energy we receive on our planet in just one hour exceeds the total amount of energy all of humanity uses in one year.
Most of our daily energy comes from the Sun. It drives our weather system, keeps us warm, powers our waterfalls, solar collectors, wind turbines, and it concentrates its energy in food that we eat and feed to our domestic animals. Without the only continuously operating hydrogen-powered nuclear fusion reactor within 4.36 light years, everything we know would grind to a complete halt.
Light of my Life
Liquid hydrogen is the lightest liquid. At -250° C one litre of the substance has a mass of only 67 grams, as compared to water which is 1000 grams per litre (the de facto standard). Part of the explanation for this is that hydrogen is the only element which can exist without a neutron, essentially halving its mass.
It IS Rocket Science
This is also why it makes such a great rocket fuel. The U.S. Space Shuttle used hydrogen and oxygen to power its main engines in a ratio of 1:6 by mass, respectively. The external tank held 106,000 kg of hydrogen and 629,000 kilograms of oxygen. The difference in volume is almost reversed, with the heavier oxygen being only 553,000 litres, but the lighter hydrogen requiring 1,497,000 litres of space. Consequently, the exhaust gas of the space shuttle main engines was environmentally friendly steam.
Solid hydrogen is also the lightest solid at only 86 grams per cubic litre. If we could actually manufacture that much, it would take almost 12 litres of solid hydrogen to attain a mass of one kilogram. Experiments in 2016 at 325 GPa (gigapascals) produced Phase V hydrogen between diamond anvils, but at that pressure the gap between the anvils was too small to get a conductor inside to see if it conducted electricity and if it was therefore a semimetal.
One GPa is equal to 9,870 Earth atmospheres. 325 GPa would equal 3,207,500 atmospheres. In one experiment they believe they reached 388 GPa (3,829,560 atmospheres). They hope to attain ~425 GPa in the next iteration and create a true metal of hydrogen.
It is not certain, of course, but it is theorized that hydrogen can be a true metal, but only at pressures normally found at a planetary core. Experiments are being undertaken to hopefully find the VI (sixth) phase of hydrogen, which should be metallic.
If they manage to create it, scientists suspect that it will be a superconductor at room temperature, or possibly exist as a superfluid that defies gravity. If it retains both states of superfluid and superconductor, which scientists have put forward as a possibility, they will have a completely unknown substance with contradictory properties on their hands. Superconductors conduct, naturally; superfluids are insulators. What will we get?
It’s Everywhere
Hydrogen is about 11% of everything biological. Naturally that includes water which accounts for a large percentage of its presence, but it also includes fats, proteins, starches, sugars, or just about any other biological material you can name.
We actually go out of our way to add hydrogen to certain foods. Rendered fats from meat processing were treated with hydrogen, or hydrogenated. This turned a liquid fat into a solid and it could be used to make flakey, non-elastic pastry in the form of shortening.
Later, in the mid-20th century, we began hydrogenating perfectly clear vegetable oils, and they had the advantage of not requiring refrigeration, in a time when refrigerators were rare. The Crisco /Cookeen/Copha generation was born.
Just For Fun
If you want to flummox your friends you can refer to hydrogen by its less common name protium (sometimes specified as 1H), named thusly because it has one proton and no neutron. In the rare case where hydrogen obtains a neutron, we then call it deuterium (2H), and in an even rarer instance, if it acquires two neutrons, that isotope is called tritium (3H).
Keychain Amusement
Tritium is radioactive to a very small degree, and is often sold as semi-permanent light source to hang on your key chain. Its specific radioactivity is so low that the glass vial that contains the tritium is more than adequate to shield the radiation. It does make it easier to find your keys in the dark though, and the light in the vial is likely to last for a decade or more (tritium has half-life of more than 12 years) before it starts to fade.
No Small Matter
The only antimatter we have ever created was made at CERN, home of the Large Hadron Collider (LHC), and it was anti-hydrogen. We could make that because all it requires is an antiproton and a positron, which are reasonably easy to come by in a particle accelerator (albeit somewhat expensive to make). The reasonably small sample was maintained for 17 minutes.
Quite Illuminating
Before we had electric street lamps, night time illumination was most often provided by burning hydrogen gas. There were alternatives, of course, such as carbon monoxide, acetylene, methane, natural gas, and coal gas, but hydrogen was relatively easy to produce.
The Takeaway
Don’t worry about running out of hydrogen. We have a lifetime supply right here on our planet. When it floats up to the sky it doesn’t necessarily escape. Molecular hydrogen, H2, on its way through the ozone layer, often encounters monatomic oxygen, and joins with it, creating a heavy molecule of water that eventually makes its way back down to Earth.
Just 5 kg of hydrogen will let you cover more than 500 kilometres/300 miles in a full size car. Hydrogen is currently selling at $10 per kilogram but as the infrastructure builds up, that is expected to drop.
Household fuel cells are available from a limited number of respectable manufacturers now. They all have their good and bad points. Some will only run on purified hydrogen; some will not operate below freezing temperatures; some fuel cells can operate on a combination of methanol and water which is far easier to acquire them pure hydrogen. Others can run on propane or natural gas if you add a chemical reformer to the system.
Check out the options before you jump on board, and remember the technology is always changing. Someone might invent a system that is just perfect for you, if it doesn’t exist already. Keep aware of developments and one day soon you can be a 100% green energy user.
 The Toyota Mirai is a hydrogen fuel cell vehicle that was first introduced to the market in 2014. It is a groundbreaking car that has the potential to revolutionize the way we think about transportation, particularly in terms of reducing greenhouse gas emissions. In this article, we will explore the technology behind the Toyota Mirai and how it works to provide a sustainable and efficient mode of transportation.
The Toyota Mirai is a hydrogen fuel cell vehicle that was first introduced to the market in 2014. It is a groundbreaking car that has the potential to revolutionize the way we think about transportation, particularly in terms of reducing greenhouse gas emissions. In this article, we will explore the technology behind the Toyota Mirai and how it works to provide a sustainable and efficient mode of transportation. It has been more than 50 years now since General Motors unveiled the first hydrogen car. An engine running entirely on clean energy? Even more, outputting drinkable water? A deal almost too good to be true. However, it is also too good to give up on easily. Still, in the last few years, electric vehicles (EVs) have taken the “green” market almost on its entire. So, who killed the hydrogen car?
It has been more than 50 years now since General Motors unveiled the first hydrogen car. An engine running entirely on clean energy? Even more, outputting drinkable water? A deal almost too good to be true. However, it is also too good to give up on easily. Still, in the last few years, electric vehicles (EVs) have taken the “green” market almost on its entire. So, who killed the hydrogen car? The lighter-than-air craft
The lighter-than-air craft  Actually, there’s more stored energy or explosive power in a car’s tank of gasoline than there is a tank of hydrogen. More importantly, gasoline splashes, and pools, and volatises (evaporates) whereas Hydrogen goes straight up.
Actually, there’s more stored energy or explosive power in a car’s tank of gasoline than there is a tank of hydrogen. More importantly, gasoline splashes, and pools, and volatises (evaporates) whereas Hydrogen goes straight up. Nowadays of course we all use the standard notation pH, so that we make consistent references and avoid confusion. So, what are we discussing when the pH symbol is bandied about?
Nowadays of course we all use the standard notation pH, so that we make consistent references and avoid confusion. So, what are we discussing when the pH symbol is bandied about?
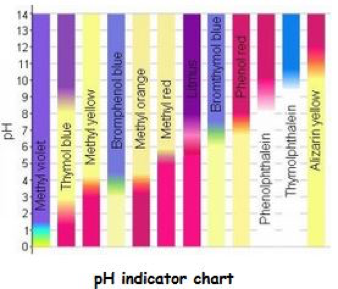 This chart gives an idea of chromatic responsiveness of several reagents. Of course there are others that are not included here. The centre strip that is half red and half purple shows the original litmus responsiveness in comparison to the greater accuracy of 11 other types.
This chart gives an idea of chromatic responsiveness of several reagents. Of course there are others that are not included here. The centre strip that is half red and half purple shows the original litmus responsiveness in comparison to the greater accuracy of 11 other types.